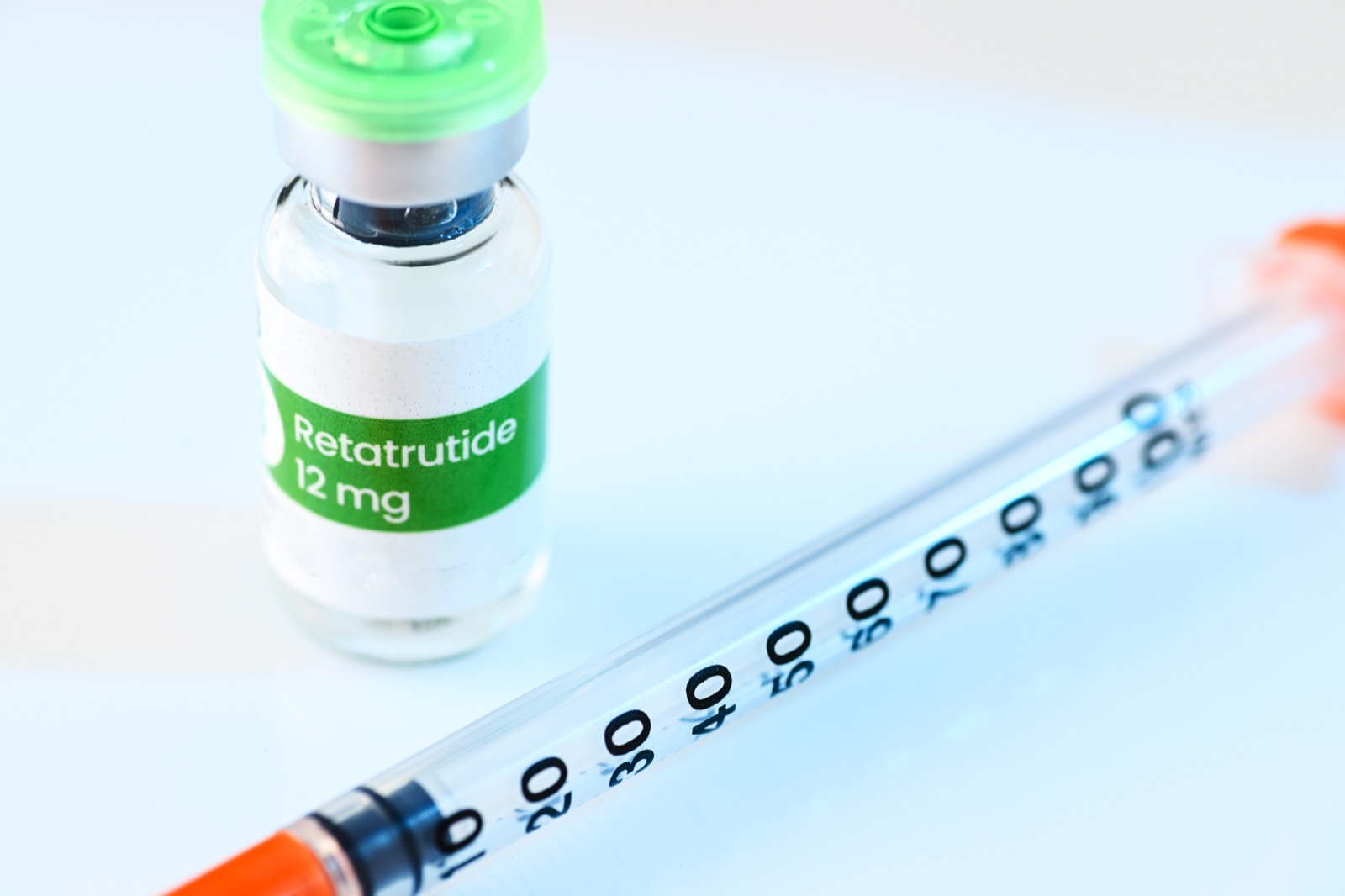
“For Research Use Only”
Every gray-market peptide vendor displays some version of this disclaimer. It is the foundational legal fiction of the entire industry. And the FDA has explicitly stated it does not work.
When a website sells vials of semaglutide alongside dosing charts, reconstitution instructions, injection guides, and before/after testimonials, the “research use only” disclaimer is legally void. The FDA has ruled that providing dosing instructions constitutes evidence of intent to sell for human use, regardless of any disclaimer text.